
Chromosomal Microarray Analysis (CMA)
Uncovering Chromosomal Abnormalities
Inquire Now
Learn more about Cordlife Chromosomal Microarray Analysis (CMA)
What is a CMA test?
Chromosomal microarray analysis (CMA) is a powerful diagnostic test that offers higher-resolution analysis of the genome compared to traditional prenatal screening tests for detecting chromosomal abnormalities.
This test detects copy number variants (CNVs), which are chromosomal abnormalities such as deletions, duplications, insertions, aneuploidy, and regions of homozygosity associated with major or rare genetic disorders.
CMA provides high-resolution insights into genetic abnormalities, aiding in the diagnosis and understanding of various medical conditions.

Why Cordlife CMA?

Comprehensive Genetic Screening
A confirmatory test that can detect chromosomal abnormalities.
Clinically Validated Technology
The sample will be analysed using SNP chromosomal microarray technology, the recommended first-tier genetic screening test for autism and intellectual disability.
Higher Resolution than Karyotyping
The resolution of the Cordlife CMA test will be greater than 200kb, as opposed to 5-10 Mb in karyotyping.

Internationally Certified Laboratory
The testing will be carried out in South korea by a CAP accredited and CLIA certified laboratory.

Turnaround Time
Your test results will be shared with your doctor within 12 to 14 days.

Complimentary Genetic Counseling
Patients who receive a positive result will receive a free genetic counseling session from our qualified genetic counselor.
Who should undergo the Cordlife CMA Test?
Cordlife CMA is suitable for the following group of patients:
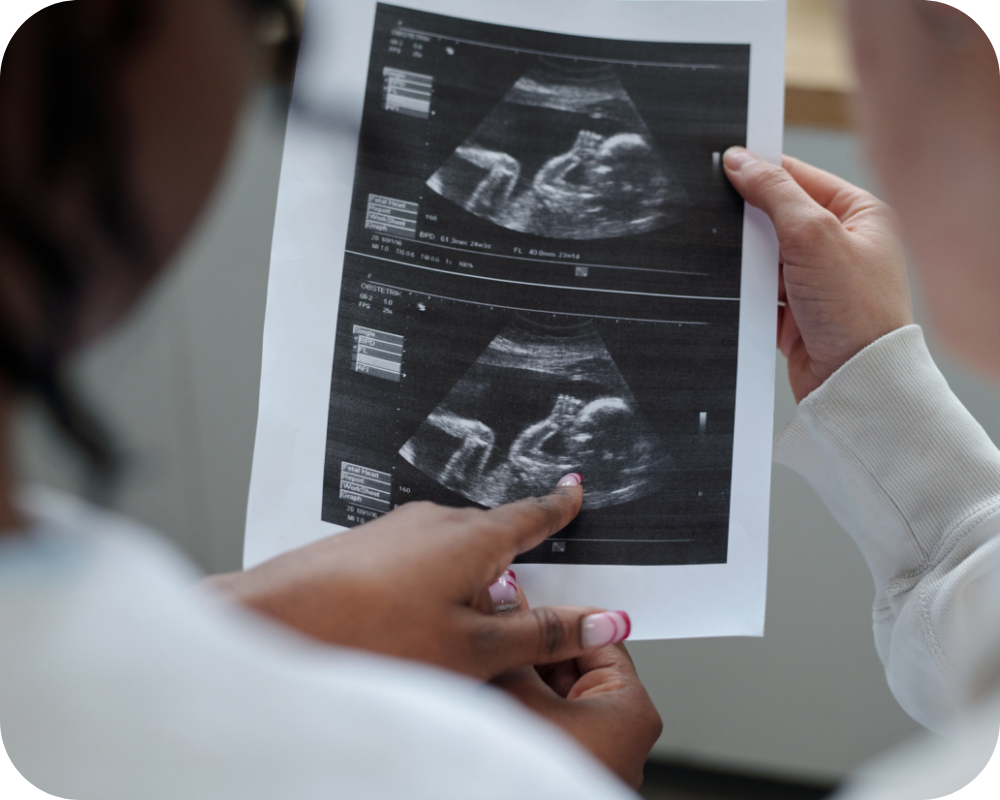
Potential fetal abnormalities revealed by ultrasound examination (including single-system or multi-system structural abnormalities)
Sample required: Amniotic Fluid

Patients with high risk Non-Invasive Prenatal Test (NIPT) result
Sample required: Amniotic Fluid

Patients with previous history of baby with congenital abnormalities
Sample required: Whole Blood

Couples encountering recurrent miscarriages
Sample required: Whole Blood
CMA compared to traditional karyotyping
Able to detect more
pathogenic CNVs
CMA testing can detect approximately 4% of pathogenic copy number variants (CNVs) that are not visible through karyotyping.1
Proven higher
diagnostic yield
According to 33 published studies involving 21,698 patients, CMA testing exhibits a diagnostic yield of 15% to 20%, compared to 3% for karyotyping.2

Higher
Resolution
The resolution of CMA testing exceeds 200kb, in contrast to the 5-10Mb resolution of karyotyping.

Recognised as a first-tier diagnostic test
CMA testing has superseded karyotyping due to its higher sensitivity and specificity.3
Find out more:
References:
- Cicatiello R, Pignataro P, izzo A, et al. Chromosomal microarray analysis versus karyotyping in fetuses with increased nuchal translucency. Med Sci (Basel). 2019;7(3):40.
- Miller DT, Adam MP, Aradhya S, et al. Consensus statement: Chromosomal microarray is a first-tier clinical diagnostic test for individuals with developmental disabilities or congenital anomalies. Am J Hum Genet. 2010;86(5):749-764.
- Martin CL, Ledbetter DH, Chromosomal microarray testing for children with unexplained neurodevelopmental disorders. JAMA. 2017;317(24):2545-2546.
